The Power of Wearables: What Your Device Can (and Can’t) Tell You About Autonomic Health

Wearable devices have changed how people engage with their health.
For the first time, millions of people can see their heart rate, sleep patterns, activity levels, and recovery trends in near real time. This visibility has sparked an important shift: people are no longer satisfied with vague explanations when they feel unwell. They want data.
But not all data is equally informative.
When it comes to autonomic health, understanding what wearables are actually measuring—and what they fundamentally cannot—is essential.
What wearables are really measuring
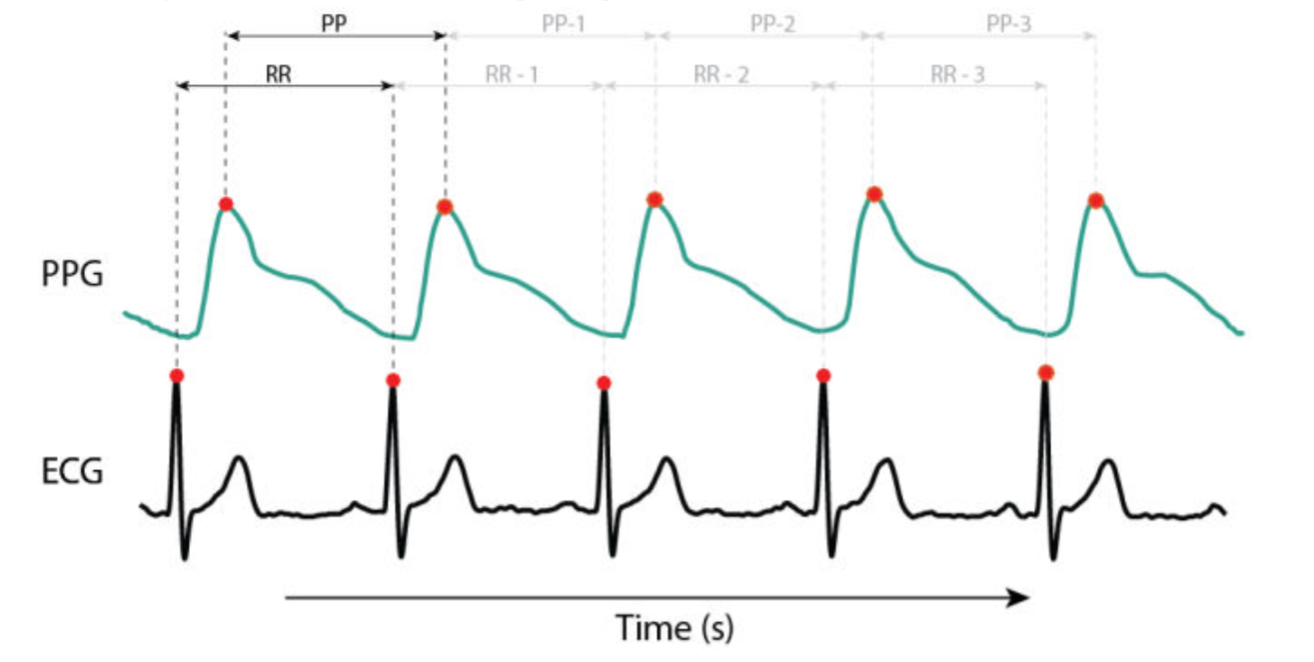
Most consumer wearables do not measure the heart’s electrical activity directly.
Instead, they rely on photoplethysmography (PPG), an optical method that detects changes in blood volume as pulse waves move through the tissue. Clinical autonomic assessment, by contrast, relies on electrocardiography (ECG), which detects the heart’s electrical R-waves with millisecond precision.
This distinction matters.
The peak of a pulse wave is rounded and variable, whereas the electrical R-wave is sharp and precisely defined. When the exact timing of each heartbeat cannot be identified reliably, calculations that depend on beat-to-beat precision—such as heart rate variability—become less accurate.
Wearables are not wrong. They are simply measuring a different signal than what autonomic physiology requires.
Why sampling rate and signal quality matter
Autonomic regulation unfolds on very short timescales.
To accurately assess sympathetic and parasympathetic activity, cardiac signals must be sampled at high frequencies, often hundreds of times per second. Many consumer wearables sample more slowly to preserve battery life and improve comfort.
Lower sampling rates introduce small timing errors, known as jitter. These errors may seem trivial, but they disproportionately distort the high-frequency components of heart rate variability that reflect parasympathetic (vagal) activity.
In other words, the data may look smooth and clean, while critical regulatory information has already been lost.
The “one number, two systems” problem
The autonomic nervous system has two independent branches: sympathetic and parasympathetic.
Many wearable metrics—such as readiness, recovery, or stress scores—attempt to summarize this two-branch system using a single composite number, often derived from heart rate variability alone.
Physiologically, this is insufficient.
Without an independent measure of respiration, it is mathematically impossible to separate sympathetic activation from parasympathetic withdrawal. As a result, most wearable-derived scores are mixed measures. They can indicate that something has changed, but they cannot explain which regulatory pathway is responsible.
This is why wearable data often raises questions rather than answering them.
Why resting data misses regulatory failure
Most wearables collect data passively, during sleep or quiet wakefulness.
But the autonomic nervous system is designed to maintain stability at rest—even when it is impaired. Dysfunction often reveals itself only when the system is challenged.
Standing up. Breathing deeply. Recovering from exertion.
Failure patterns such as sympathetic withdrawal or dynamic parasympathetic excess frequently remain invisible during steady-state monitoring. They emerge only when the system is forced to adapt.
This is why people can have normal-looking wearable data while experiencing severe dizziness, fatigue, or exercise intolerance in daily life.
The respiratory confound
Breathing profoundly shapes autonomic signals.
Two people can have identical heart rate variability values while being in very different physiological states if their breathing patterns differ. Slow breathing can shift parasympathetic activity into frequency ranges that many algorithms misinterpret as sympathetic stress.
The result is a false signal: relaxation appears as strain, or strain appears masked.
Without accounting for respiration, HRV-based interpretations are inherently limited.
When artifacts masquerade as health
Wearables must contend with motion, poor contact, skipped beats, and irregular rhythms.
If these artifacts are not rigorously detected and corrected, they can be misclassified as healthy variability. In some cases, a stressed or unstable heart can appear deceptively “variable” and therefore healthy.
Clinical autonomic analysis relies on advanced signal-processing techniques designed to distinguish true physiological variability from noise. Many consumer algorithms are optimized for trend detection, not physiological separation.
This is another reason wearables are better at awareness than diagnosis.
Monitoring versus physiological testing
There is a fundamental difference between monitoring and testing.
Monitoring observes what happens over time. Testing measures how the system responds to a defined challenge.
Wearables excel at monitoring. They can capture long-term trends and flag irregular events.
Autonomic testing, however, evaluates response and recovery under controlled conditions. It assesses how quickly the nervous system adapts, compensates, and returns to baseline. This distinction is critical for identifying regulatory dysfunction.
One provides context. The other provides explanation.
How wearables fit into the future of autonomic care
Wearables are not the endpoint. They are the infrastructure.
High-quality devices can support autonomic testing by supplying reliable cardiac data, longitudinal context, and at-home accessibility. But interpretation must be grounded in physiology, not scores alone.
At Autonomic Health, the goal is to bridge this gap by combining compatible wearable data with validated autonomic testing protocols, transforming raw signals into meaningful insight about regulation, adaptability, and recovery.
Choosing the right expectations
If you use a wearable and feel confused by your data, you are not failing to interpret it correctly.
You are encountering its limits.
Wearables are powerful tools for awareness and habit formation. They were never designed to diagnose autonomic dysfunction or explain complex, multi-system symptoms.
Understanding what your device can and cannot tell you prevents frustration—and opens the door to deeper insight.
The next step beyond tracking
The future of health is not more dashboards. It is better interpretation.
Wearables helped people see that the nervous system matters. Autonomic testing makes it possible to understand how it functions.
Tracking is powerful. Understanding is transformative.
Autonomic testing is coming soon
At-home autonomic testing is currently in development.
You can sign up on our website to be notified when testing becomes available and learn how your wearable data fits into a clearer picture of nervous system regulation.
Selected references (for those who want to go deeper)
Freeman R. Assessment of cardiovascular autonomic function. Clinical Neurophysiology (2006).
Goldberger JJ, Arora R, Buckley U, Shivkumar K. Autonomic Nervous System Dysfunction: JACC Focus Seminar.Journal of the American College of Cardiology (2019).
Aysin B, Aysin E, Colombo J. Separation of sympathetic and parasympathetic components of heart rate variability using advanced signal processing. IEEE Engineering in Medicine and Biology Conference (2007).
Colombo J, Arora RR, DePace NL, Vinik AI. Clinical Autonomic Dysfunction: Measurement, Indications, Therapies, and Outcomes. Springer (2014).
Malik M, et al. Heart rate variability: Standards of measurement, physiological interpretation, and clinical use.Circulation (1996).
Shaffer F, Ginsberg JP. An overview of heart rate variability metrics and norms. Frontiers in Public Health(2017).